Biomaterials Hydroxyapatite

Biomaterials Hydroxyapatite
Inorganic materials for the biomaterials field
Hydroxyapatite
- Hydroxyapatite is basic calcium phosphate and exists naturally as a major component of bones and teeth and as an ore in nature.
- Hydroxyapatite shows high biocompatibility and is utilized in biomaterials such as artificial bones, dental implants, and coating materials for artificial joints.
- Tomita Pharmeceuticals’ hydroxyapatite conforms to ASTM* standards, and in addition to meeting standards for quality and impurities as raw material for medical equipment, its physical properties are strictly controlled according to the requirements of each application.
- Since impurities are controlled and managed during production, there are few elemental and crystalline structural impurities, resulting in high purity.
- It therefore inherently has a high degree of whiteness, making it ideal for products with a white coating.
*ASTM standards are developed and published by ASTM International (formerly known as the American Society for Testing and Materials), the world’s largest standards organization.
- Chemical name: Hydroxyapatite (tricalcium phosphate)
- Chemical composition: Ca10(PO4)6(OH)2 CAS No. 1306-06-5
| Product Name | Use | Characterstics | |
|---|---|---|---|
| Hydroxyapatite C | For industrial use | Abrasives, materials for plasma thermal spraying, raw materials for biomaterials (artificial bone, dental implants, bone filler) |
|
| Hydroxyapatite HD | |||
| Hydroxyapatite FP | |||
Hydroxyapatite for Thermal Spraying
Our hydroxyapatite conforms to ISO13779-6. Since medical devices such as implants exist in the body for a long period of time, elemental impurities and analogous substances in their raw materials must be strictly controlled.
We strictly control hydroxyapatite from raw materials to manufacturing and quality inspection, and can provide products that qualify as raw materials for medical equipment as well as conforming to ISO.
Physical properties
| Test case | Example of Analysis | |||
|---|---|---|---|---|
| Hydroxyapatite C | Hydroxyapatite HD | Hydroxyapatite FP | ||
| Bulk density(ISO 3923-1) | 0.5 g/mL | 0.8 g/mL | 1.0 g/mL | |
| Particle size distribution | D10 | 57 μm | 57 μm | 15 μm |
| D50 | 91 μm | 93 μm | 28 μm | |
| D90 | 150 μm | 156 μm | 48 μm | |
SEM image
| Hydroxyapatite C | Hydroxyapatite HD | Hydroxyapatite FP |
|---|---|---|
 |
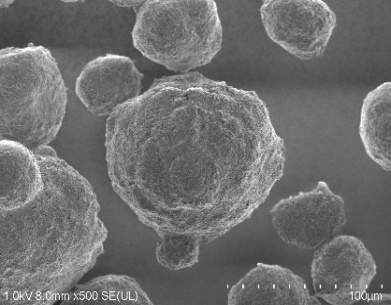 |
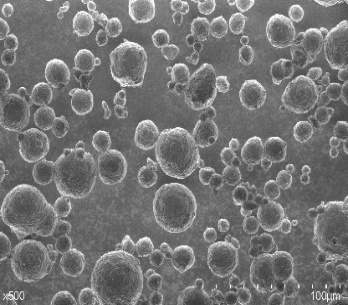 |
Example of application as a coating material
We performed plasma thermal spraying experiments to confirm the coating characteristics of our hydroxyapatite on Poly Ether Ether Ketone (PEEK), which is used as a base material for implants.
Test conditions
| Test Case | Conditions | ||
|---|---|---|---|
| Thermal Spraying Method | Atmosphere Plasma Spray | ||
| Base Material | PEEK | ||
| Coating Material |
|
||
| Blasting conditions | Media | Al2O3 (WA♯46) | |
| Pressure | 0.5 MPa | ||
| Distance | 150 mm | ||
| Thermal Spray Equipment |
|
||
| Working gas | Ar | SLPM | 30 |
| He | SLPM | 150 | |
| Carrier gas | N2 | SLPM | 4 |
| Current value | A | 500 | |
| Spray distance | mm | 100 | |
Result
Microscopic image of coated film
| Before coating | After coating |
|---|---|
 |
 |
Cross-sectional image of coated film
| Magnification | Hydroxyapatite FP | Competitor Product |
|---|---|---|
| ×200 |  |
 |
Our hydroxyapatite FP formed a film on the PEEK surface in a uniformly melted state.
In contrast, the competitor product failed to form a film.
Characteristics of Coating Film
| Test case | Result | ||
|---|---|---|---|
| Purity | Crystallinity | 63% | |
| Impurity Phase | α-TCP | 1.40% | |
| β-TCP | 1.60% | ||
| TTCP | 1.00% | ||
| CaO | 0.20% | ||
| Ca/P ratio | 1.67 | ||
| Characteristics | Vickers hardness | 244 (HV0.3) | |
| Abrasion | 4.2 mg | ||
| Film thickness | 170 um | ||
| Surface roughness (Ra) | 4.1 um | ||
Hydroxyapatite FPs showed little decomposition to impurity phase after thermal spraying and formed a high-strength film.
Therefore, it showed desirable properties as a surface coating material for medical equipment.
Contact Us
We are happy to answer any questions or concerns you may have about our products or our company.
Please feel free to contact us.
